 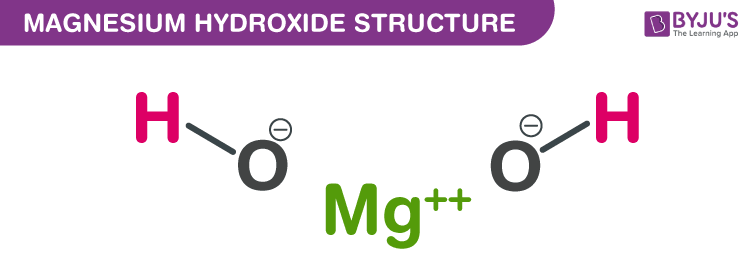
High doses might cause diarrhoea and decrease the body's potassium supply, which can result in muscular cramps. Solution: Atomic mass of Mg 24.3 amu Atomic mass of H 1.008 amu Atomic mass View the full answer. The osmotic force of magnesia functions as a laxative, drawing fluids from the body. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. It can also be used as a laxative to help with constipation. Magnesium is a chemical element with the symbol Mg and atomic number 12. These antacids are used to neutralise stomach acid and alleviate indigestion and heartburn. Magnesium hydroxide is available in a variety of forms for medical usage, including chewable tablets, capsules, powder, and liquid solutions, which are occasionally flavoured. The molar mass is stated in g/mol and is defined as the mass of a particular substance divided by the quantity of that substance. The molecular mass and relative molecular mass are not the same as the molar mass, but they are linked. The related quantity relative molecular mass is a unitless ratio of a molecule's mass to the unified atomic mass unit (also known as the dalton) as defined by IUPAC. Because they contain various isotopes of an element, multiple molecules of the same chemical may have different molecular weights. When combined with water (H 2 O), magnesia forms magnesium hydroxide (Mg (OH) 2. Magnesium oxide is used in some antacids, in making crucibles and insulating materials, in refining some metals from their ores and in some types of cements. Hint: The mass of a molecule is measured in daltons and is called molecular mass (m) (Da or u). Magnesium oxide (MgO), also known as magnesia, is the second most abundant compound in the earth's crust.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
